Proteins in cell membranes play a key role in many diseases and therapies. Pontus Gourdon is creating high-resolution images of proteins in order to understand them in detail, and figure out how they can be modified to attain the best function.
Pontus Gourdon
PhD, membrane protein structural biology
Wallenberg Academy Fellow, prolongation grant 2020
Institution:
Lund University
Research field:
3D imaging of membrane proteins
One-third of the 30,000 or so proteins synthesized by the human body are membrane proteins. They operate as channels, pumps and doors in the cell membrane, and are responsible for communication between the inside and outside of the cell. More than half of all medicines target membrane proteins.
Gourdon, who is a Wallenberg Academy Fellow at Lund University, is working on three-dimensional, high-resolution images of the proteins, and films showing how they work.
“All films are in fact a series of still images. If we have enough images of different phases of a process, we can put them together to make a film. It’s tricky working out the order in which to arrange them, but it’s really intriguing.”
Gourdon’s research field – membrane protein structural biology – has undergone a major technology transition over just a few years. When he was first chosen as a Wallenberg Academy Fellow in 2015, the dominant technology was X-ray crystallography. This entails mixing proteins with chemicals that induce them to form crystals. With a little luck a good shape is obtained, and X-rays can be used to reveal the three-dimensional structure. In recent years another technology – cryo-electron microscopy (cryo-EM) – has increasingly taken over. It involves use of a frozen solution, avoiding the challenging step of crystallization. Originally, inferior resolution was obtained using this technique, but that quickly changed. These days Gourdon relies almost exclusively on cryo-EM, which his group was embracing early. With this technique, they have succeeded in imaging a human membrane protein, a channel for chloride ions, whose structure was previously unknown.
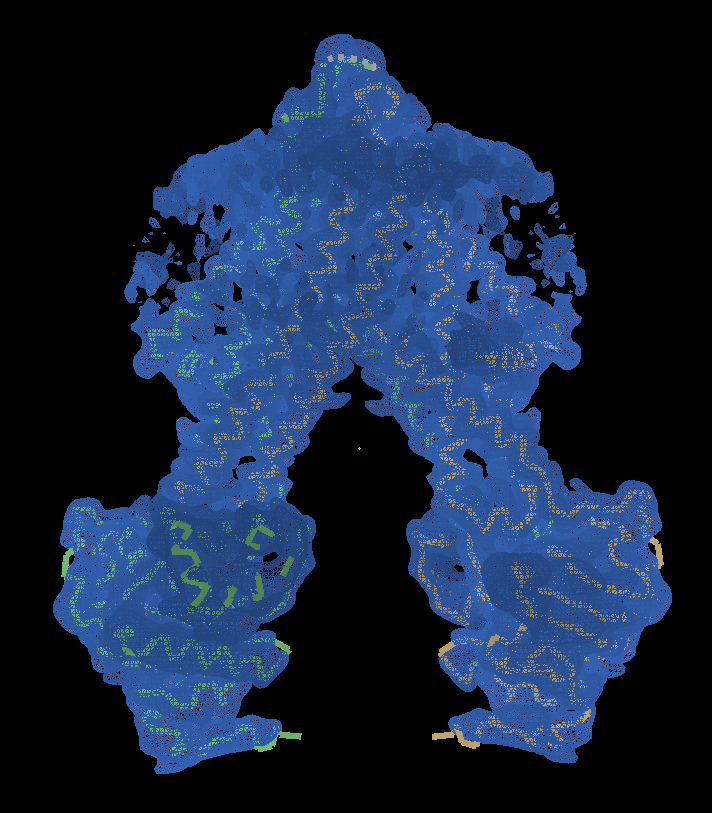
Imaging the environment, not just the protein
Gourdon wants to start imaging the environment surrounding the proteins, instead of just one protein at a time.
“The new technique enables us to determine the myriad structures in a fragment of membrane. It’s an extraordinarily complex environment, with other proteins, ions and lipids combining to control and regulate how the protein works. I want to get closer to reality and find out how it works.”
He is focusing on a certain family of proteins, known as P-type ATPases. ATP are molecules that serve as a source of energy. The protein family comprises ATP-driven membrane pumps that counteract the equalization of substances that otherwise occurs naturally between the inside and outside of the cell. They make sure that the concentrations or pH of various substances differ inside the cell from those outside. One of these pumps transports copper ions in human as well as bacterial cells. Gourdon wants to find out whether it can be used to develop a new kind of antibiotic.
“In some bacterial infections the body tries to fight the intruder by increasing the concentration of copper. Copper is toxic in high doses, and the bacterium protects itself by getting rid of the copper ions via a pump of this kind. Our idea is to try to block it.”
Blocking the pump could also inhibit the copper pumps in human cells. The aim is to avoid this by designing a molecule specifically for the bacterial pumps, thereby avoiding side-effects.
“But even if the human pump were blocked for a short time, it might not be a problem, since a bacterial infection can be dealt with in a week or two.”
Potential for more efficient drug development
In-depth knowledge of protein forms can also be used in the development of other therapeutics. Drugs often act by inhibiting a given protein, and their efficacy depends partly on how strong the binding to the protein is. If the “pocket” to which the drug attaches is completely filled, the binding will be stronger. Gourdon’s images can show whether the drug needs to be designed a little differently to create an even better fit.
“Modifications like this have been made before, but it’s been done more in a kind of black box – molecular changes made at random and tested to see what works. Knowledge of these structures may enable us to develop drugs more efficiently and rationally,” Gourdon says.
“Without my Wallenberg Academy Fellow grant it would have been difficult to make this technological transition. It’s played a vital role in the results we’re now seeing.”
Over the past few years his life as a researcher has changed quite considerably. As a team leader he spends much less time in the lab than he used to, and he sometimes misses it. But he is delighted that the team is now making more rapid progress.
“We’re carrying out more complex experiments, and obtaining much more information as a basis for each scientific article than we used to. In that sense the work is infinitely more enjoyable. But competition in the field is growing constantly, which can be stressful at times – a researcher always wants to be the first to make a discovery.”
Text Lisa Kirsebom
Translation Maxwell Arding
Photo Ping Li, Pontus Gourdon






