Huntington’s disease is caused by a single gene. All carriers are affected, and there is no cure or medication to slow the disease. Wallenberg Clinical Scholar Åsa Petersén is focusing on early signs of mental impairment with the aim of finding new pathways to treatment.
Åsa Petersén
Consultant in Psychiatry and Professor of Neuroscience
Wallenberg Clinical Scholar 2020
Institution:
Lund University
Research field:
Huntington’s disease and its effects on the hypothalamus
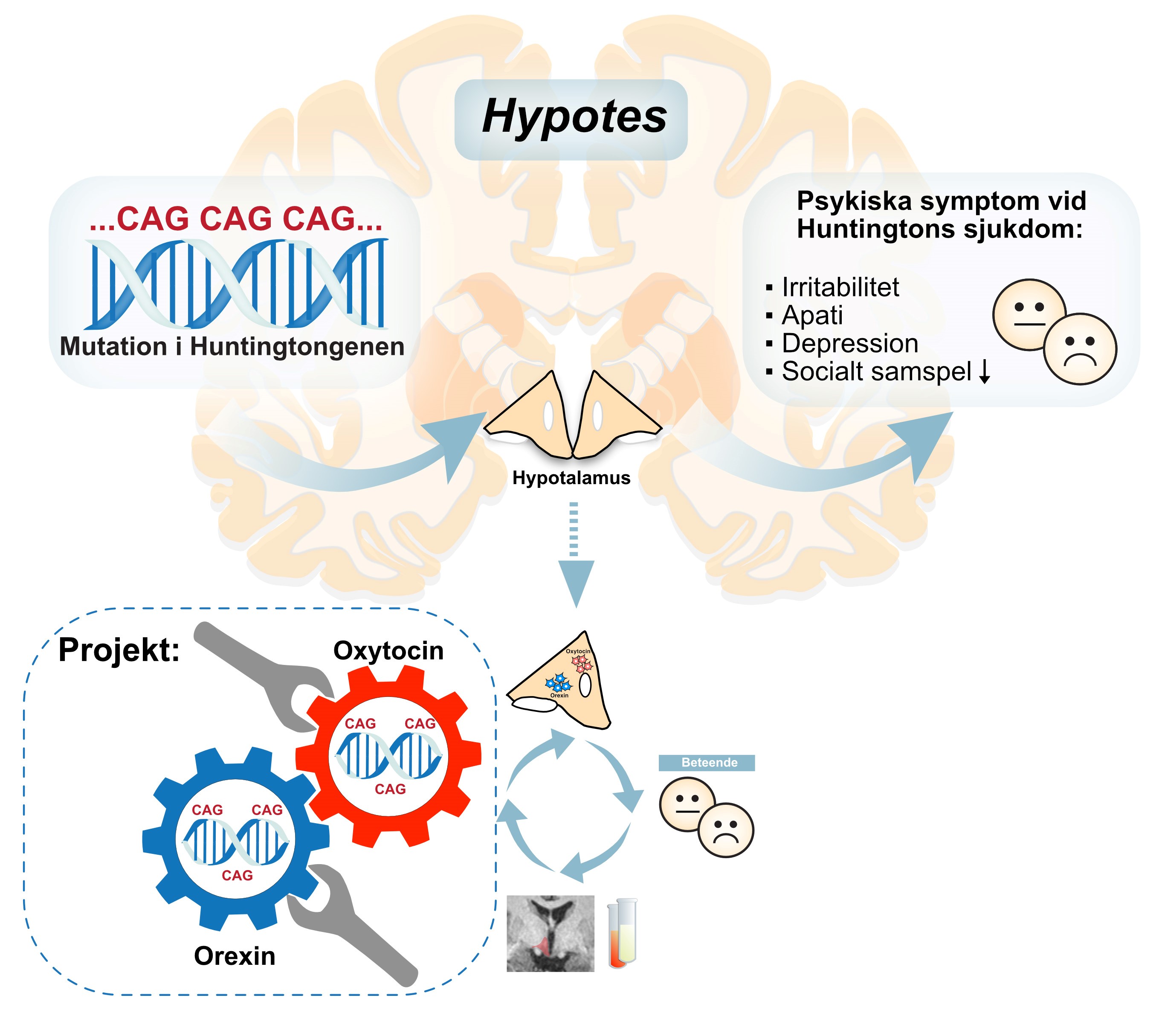
Huntington’s disease occurs when a microscopic strand of DNA code is repeated too many times by mistake. A person with these repeats in their gene coding always develop the disease, and their offspring run a 50-percent risk of inheriting the gene. The mutant gene synthesizes an abnormal mutant form of a protein called huntingtin. The mutant protein is present in all cells, but for some reason only causes damage in the brain. Among other things, it kills certain types of neurons in a part of the brain called the basal ganglia, one of the brain’s motor centers. The cell death results in involuntary jerky movements. The symptoms become more severe over time. Huntington’s patients usually die about twenty years after diagnosis.
Åsa Petersén at Lund University has researched the disease for many years, and her findings have yielded a new insight: the defective huntingtin also causes cell death in the hypothalamus. This is the brain’s hormone center, and the damage caused there can result in anxiety, depression, irritation, disturbed sleep patterns and impaired executive functions.
“As a doctor it is clear to me that psychiatric symptoms cause the most anguish, both for the affected individuals and their families. And those symptoms appear long before motor dysfunction. If we can find out the cause, we may be able to find new approaches to treatment and stop the progress of the disease.”
First symptoms psychiatric
It was previously thought that the psychiatric problems were a reaction to the disease, but it is now clear that those problems appear long before the patients are aware of their condition. They can begin up to twenty years before impaired motor function. Petersén has also demonstrated that mice with the Huntington gene exhibit similar psychiatric symptoms as humans.
“We now know that metabolism, sleep and emotional regulation are affected early on. All these can be controlled from the hypothalamus, where we have seen changes both in MRI scans, and in brain tissue from people who have died from the disease.”
The reason that only certain cells are damaged by Huntington’s remains a mystery. Research has shown that cells in the basal ganglia die because the mutant huntingtin disrupts multiple processes, from signaling to waste disposal.
“I want to take a closer look at the hypothalamus, and study the molecular changes that occur. There are signs that the processes there are similar. We know that the cells affected are those that synthesize the hormones orexin och oxytocin.”
Orexin affects appetite and motivation, whereas oxytocin is the “happy hormone”, reducing aggression, depression and stress.
Lab experiments combined with patient studies
As a Wallenberg Clinical Scholar, Petersén is studying neurons from the hypothalamus in the laboratory, and using genetic tools to control them, so she can study in detail how mutant huntingtin impacts them and other parts of the brain. Another part of the project involves studying people with the Huntington gene to see what changes have occurred in the hypothalamus, and Petersén and her colleagues also plan clinical studies to test experimental therapies.
“We want to understand exactly what it is in the brain that causes the psychiatric symptoms, but the studies may also yield vital knowledge about what causes the same symptoms in other mental illnesses. New therapeutics approved for other diseases might also work on Huntington’s.”
A number of patient studies investigating potential new treatments for Huntington’s are currently in progress, among them gene therapies. But all of them to date target patients who already exhibit motor symptoms.
“At present we don’t really know whether the disease can be slowed at that stage, or whether treatment must begin earlier.”
“I’m delighted to have received this grant. It’s marvelous that there is a program specifically for those wholeheartedly combining research and medical practice. It’s a demanding combination, but a hugely rewarding one.”
Research as a mission
Petersén has always been interested in the workings of the brain, and was drawn to the riddle of Huntington’s disease: a known genetic mutation seen in all cells but only affecting a small proportion of them, and only causing certain symptoms. As a doctor, she sees many of the affected families in the southern Swedish health care region. This is because Huntington’s is so uncommon that patients are referred to the hospitals with the greatest expertise and experience of the disease.
“Meeting the families is a very moving experience – it has become an important part of my life. I have the privilege of trying to help them in various ways – now and by researching into potential new therapies. It has come to be my mission in life.”
As gene analysis technologies have become better and less expensive, and fetus diagnostics has become widely used, it might be imagined that Huntington’s disease is becoming less common. But more than one case in ten occurs spontaneously, without any other cases among relatives. And many of those who inherit the disease choose not to undergo a genetic test before they exhibit symptoms.
“When an older relative is diagnosed with Huntington’s, many young people first say they want to be tested, but a year later many of them have changed their minds. What is right for one person is not right for another. It’s an incredibly difficult choice to make.”
Text Lisa Kirsebom
Translation Maxwell Arding
Photo Agata Garpenlind, Kennet Ruoma, Sanaz Gabery